AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
 Recovery after cord blood transplants is also dependent on the use of in vivo T-cell depletion as outlined below and can be on par with unmodified grafts in the absence of anti-thymocyte globulin (ATG). In general, immune recovery occurs more rapidly after unmodified graft transplants than in the setting of in vivo or ex vivo T-cell depletion. As noted above several factors influence immune recovery after allogeneic HSCT. When comparing immune recovery across graft sources and transplant approaches, the available data suggest that immune recovery occurs most rapidly in recipients of autologous grafts. In the first months after a HSCT peripheral expansion of the donor T cells is the dominant mechanism for T-cell recovery (except in the recipients of a T-cell depleted allograft) and results in particular in clonal expansion of CD8+ T cells with a limited repertoire. Thymopoiesis occurs as a crosstalk between developing thymocytes and the stroma, which includes dendritic cells, macrophages, fibroblasts, endothelial cells, B cells, and thymic epithelial cells (TECs). Lymphoid precursors traffic from the BM to the thymus and undergo a complex process, including proliferation, differentiation, and positive and negative selection resulting in the export of functional CD4 and CD8 T cells. 6, 7 The thymus is the primary site for the development of T cells. 6, 7 Post-transplant T-cell recovery can occur through 2 mechanisms: (1) survival and peripheral expansion of infused donor (memory) T cells, and (2) de novo generation of donor T cells in the thymus from donor hematopoietic precursors. 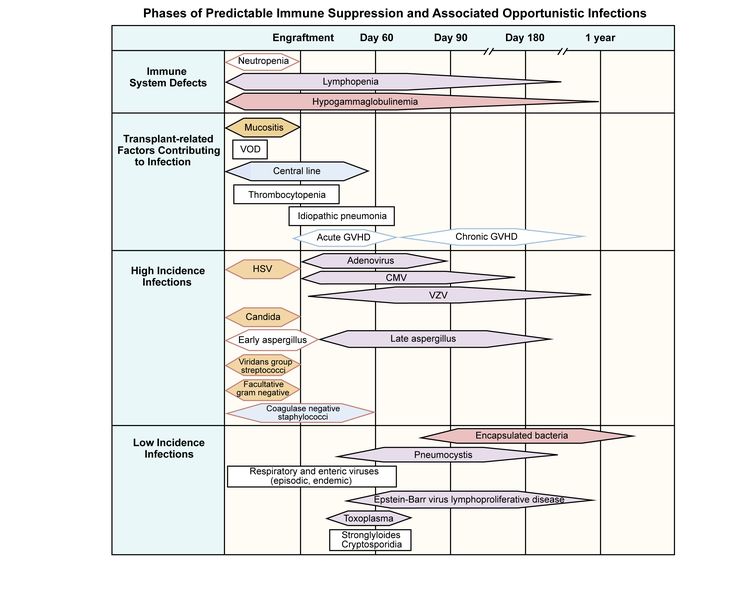
5 In contrast, adaptive immunity, which consists of cellular (T lymphocytes) and humoral (B lymphocytes) immunity, takes 1-2 years to recover and a significant number of patients will incur even longer-lasting deficits. HSCT VOD/SOS defibrotide elastometry liver stiffness measurement.Ĭopyright © 2020 Bonifazi, Barbato, Ravaioli, Sessa, Defrancesco, Arpinati, Cavo and Colecchia.Innate immunity recovers in the first months after HSCT: first monocytes, followed by granulocytes and natural killer cells. During the last years, decreased mortality associated to VOD/SOS has been reported being it attributable to a better intensive and multidisciplinary approach. Because of its severity, particularly in VOD with associated multiorgan disease, prophylaxis approaches are under investigation. Treatment includes supportive care, intensive management, and specific drug therapy with defibrotide. Risk factors associated with the onset of VOD and diagnostic tools have been recently updated both in the pediatric and adult settings and here are reported. The injury of the sinusoidal endothelial cells with loss of wall integrity and sinusoidal obstruction is the basis of development of postsinusoidal portal hypertension responsible for clinical syndrome. We review pathogenesis, clinical appearance and diagnostic criteria, risk factors, prophylaxis, and treatment of the VOD occurring post-HSCT. 
Hepatic veno-occlusive disease (VOD) or sinusoidal obstruction syndrome (SOS) is a rare complication characterized by hepatomegaly, right-upper quadrant pain, jaundice, and ascites, occurring after high-dose chemotherapy, hematopoietic stem cell transplantation (HSCT) and, less commonly, other conditions. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
